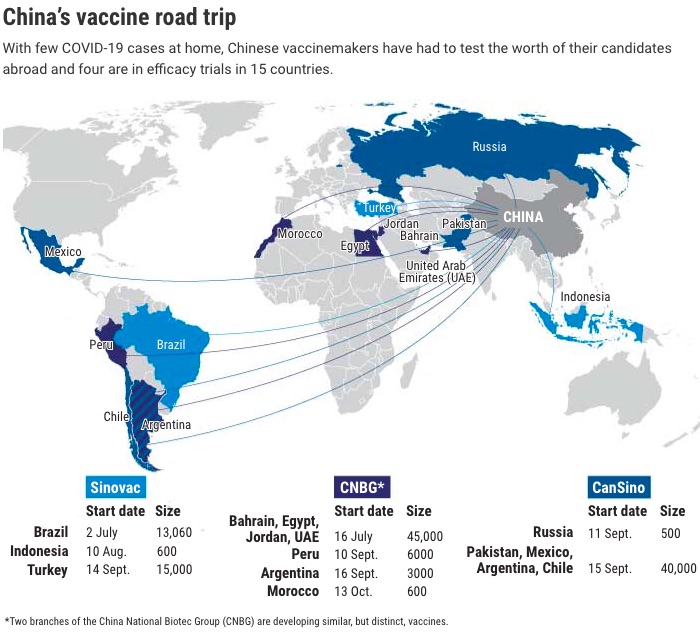
We have been saying it backwards and forwards, China is already the centre of the world and its leadership will dangerously relegate investors who remain focused on the West and fearful of the badly named emerging markets (since many are already emerging and most of the West has not yet realised it). Check if not the pre- and post-pandemic economic growth data and projections.
.
This time it is a scientific rather than an economic publication that alerts us to China's hegemony. Below we translate, summarise and comment on the magazine's article. Science by Jon Cohen, which analyses China's progress in the field of vaccination against Covid19 and the dreaded SARS-CoV-2 coronavirus. And above all, it highlights the bio-economic influence that China is achieving across the globe, as always in a stealthy, intelligent and unstoppable manner:
.
The first people in the world to receive the COVID-19 vaccine were not part of a clinical trial. No television networks or newspapers covered the historic event. No company issued a statement.
.
On 29 February 2020, less than 2 months after the world awoke to the threat of the new disease, virologist Chen Wei, a major general in the Chinese army, and six military scientists on her team squared themselves in front of a Chinese Communist Party flag and received injections of an experimental COVID-19 vaccine. Chen, a national hero for her work on Ebola vaccines, had come to the pandemic's ground zero, Wuhan, with her group from the Academy of Military Medical Sciences, in part to help create the candidate vaccine with a commercial company, CanSino Biologics. Commentators inside and outside China later questioned whether the event, which was widely reported on social media, was real. No less than the People's Daily, the Communist Party's main newspaper, labelled a photo of Chen receiving the vaccine as «#FAKENEWS». But Hou Li-Hua, a researcher at the academy working on the vaccine project, says it was «real news», an attempt to protect the hard-hit city's scientists.
.
In the US, the Trump administration's $10.8 billion Operation Warp Speed has accelerated vaccine research and development specifically for the US population, and is doing so faster than many researchers thought possible. But an equally massive effort is underway in China. CanSino and two other Chinese companies - one government-owned and the other working closely with its regulatory agency - are investing substantial resources, testing four candidates in tens of thousands of volunteers worldwide, and are likely to be just days or weeks away from announcing efficacy results from the trials, on the heels of encouraging results announced over the past month by Pfizer and BioNTech, Moderna, AstraZeneca and Oxford University, and Russia's Gamaleya Research Institute of Epidemiology and Microbiology.
.
But the low profile of those historic first injections, the military's collaboration with a «private» company and the ethically fraught decision to begin vaccinations outside of a clinical trial telegraphed that, apart from similar scale and speed, China's vaccine effort is following a very different course from that of the United States and Europe. Most major Western vaccines are based on attractive modern technologies such as genetically modified viral vectors, designer proteins and RNA fragments. By contrast, three of China's four leading vaccine candidates use a classic, tried and tested method: inactivated whole virus. An approach that dates back to the first successful influenza vaccine in the 1930s. And China's vaccine effort is weighed down by its radical success with aggressive public health measures to stop the spread of the SARS-CoV-2 coronavirus, as its measures have left China with virtually no virus on which to test vaccines, including forced isolation of cases and testing of entire cities. By contrast, the raging pandemic in the US has allowed trials there to quickly show signs of efficacy. «China crushed the coronavirus epidemic early, so they missed the opportunity to test the efficacy of their vaccines there,» says epidemiologist Ray Yip, who closely follows the development of the COVID-19 vaccine as an advisor to Bill Gates. «If they had had a lot of cases in China, they could have finished an efficacy trial earlier than other countries.
.
So China's vaccine developers have gone abroad. Although the US has excluded them from Operation Warp Speed, they have negotiated with 15 other countries on five continents. They have mounted massive trials in the Arab world - and given candidate vaccines to senior government officials there, and also conveniently cajoled the radical Bolsonaro in Brazil, where the pandemic is raging, to test a vaccine and explore its production there.
.
But China is not just looking for promising sites for clinical trials. It does not urgently need vaccines at home to combat a virus it has largely crushed, but is playing a global game by pledging to send any proven vaccines to countries that are conducting trials for its candidates, or to share the technologies behind them. «They know they don't need a vaccine to contain the epidemic in China,» says Yip. «They can take their time,» and take a much longer and more long-term view. strategic.
As seen in the photo above, Chinese company CanSino Biologics created the first COVID-19 vaccine to enter clinical trials, and by April it had advanced to a phase II study in Wuhan.
.
Yanzhong Huang, a global health specialist at both Seton Hall University and the Council on Foreign Relations, says the country is «actually using the vaccine to promote the diplomacy of foreign policy objectives». This «vaccine diplomacy», he says, contrasts sharply with Warp Speed's «vaccine nationalism» and aims to «fill the vacuum left by the US».
,
«It's a very carefully executed and thought-out strategy,» says Stephen Morrison, who directs the Center for Global Health Policy at the Center for Strategic and International Studies. «A strategic goal of the Chinese government is to achieve hegemonic influence in the next 10 years..»
.
At home, too, attitudes toward vaccines contrast with those in the United States and Europe, where distrust is high, Morrison says. To the dismay of vaccine experts abroad, hundreds of thousands of people in China have already lined up to receive experimental vaccines, even before their value and safety have been proven. «There has not been a collapse of faith and trust in science and the state,» says Morrison. «There is less fear about where this is all going.» Paradoxically, it is the Westerners, the die-hard freedom advocates who question isolation measures, restrictions on movement and the reliability of testing, who have lost the most freedom as a result of their radical confinements and perimeter closures.
.
In China, the speed with which Chen and his colleagues were able to get those first vaccines is all the more remarkable given that CanSino was arguably slow.
.
Although some COVID-19 vaccinators launched their projects the day after the SARS-CoV-2 sequence was made public on 10 January, CanSino CEO Yu Xuefeng had reservations. «We started looking into it in mid-January, but there was a hesitation,» he says. COVID-19, Yu was concerned, might be a bluff, The disease, such as severe acute respiratory syndrome (SARS), another disease caused by a coronavirus, alarmed the world in 2003 but disappeared a year later, after companies and governments had devoted many resources to developing vaccines.
.
Yu, who is originally from China, completed his PhD in microbiology at McGill University in Canada in 1997, and then stayed on, working on vaccines for almost 9 years at a Sanofi Pasteur branch there. He co-founded Canino in 2009. A team led by Chen in China helped develop his only previous product to receive approval: an Ebola vaccine based on a widespread and largely harmless virus known as adenovirus 5 (Ad5), into which they added a gene for the surface protein of the Ebola virus.
.
Yu and his team considered making an anti-COVID-19 vaccine with messenger RNA (mRNA) for the coronavirus« novel surface protein, called spike, the innovative approach taken by Pfizer and its partner BioNTech, the »winner« of the race to report preliminary efficacy data. But CanSino decided to go with what it knew, using the Ad5 vector to carry the spike gene. »I thought it was the fastest and most mature way to develop a new vaccine," says Yu.
.
Within a month, CanSino's candidate was ready for delivery to Chen and his team, and on 16 March the company launched the world's first COVID-19 vaccine trial in Wuhan to test its safety and ability to elicit immune responses. CanSino had beaten Moderna by eight hours, though a world paralysed by the vaccine race among Western companies paid little attention.
.
Several US and European contenders, including AstraZeneca, have also adopted adenoviruses to carry the target protein, with some opting for an Ad5 vector similar to CanSino's, despite several concerns about the approach. In 2007, two disastrous efficacy trials of an Ad5-based AIDS vaccine found that, for reasons still unclear, it actually increased the risk of HIV infection. The other concern is that pre-existing immunity to Ad5 may attack the vaccine vector, which may explain why, in early trials, the CanSino vaccine elicited a weaker-than-expected antibody response. «We see that there is some impact,» Yu acknowledges, «but it's not black and white.» (Preliminary efficacy data for the AstraZeneca/Oxford vaccine suggest that immunity against its adenovirus vector may have compromised that candidate's performance as well, at least in double-full-load dosing.).
The other two Chinese players, Sinovac Biotech and China National Biotec Group (CNBG) - a subsidiary of one of the world's largest vaccine manufacturers, state-owned Sinopharm - are taking a different approach: vaccinating people with the whole, «killed» virus. This does not require sophisticated protein or RNA design or genetic engineering: Scientists simply inactivate the virus with a chemical (beta propiolactone) and mix it with an adjuvant that effectively puts the immune system on full alert. In theory, these vaccines can produce broader antibody and T-cell responses, because they contain the full set of viral proteins, rather than just one. And unlike mRNA vaccines, which have to be stored at sub-zero temperatures, inactivated viruses require no more than the ordinary refrigeration of any household fridge.
.
But many scientists see inactivated virus vaccines as outdated, difficult to manufacture in large quantities and potentially dangerous. Warp Speed rejected the approach outright. «I don't think the inactivated vaccine is a good idea,» says Moncef Slaoui, chief scientist at Warp Speed.
.
A major concern is that inactivated SARS-CoV-2 vaccines could trigger a more severe disease, known as «enhanced respiratory disease», in immunised people who do become infected. Basically, if a vaccine triggers ineffective antibodies, they can form immune complexes that clog the lungs. This happened with a respiratory syncytial virus vaccine given to children in the 1960s, and in animal experiments with vaccines against SARS and another coronavirus disease, Middle East respiratory syndrome. The prospect of growing large batches of virus before killing them also poses problems; twice in the last five years, live poliovirus has escaped from European plants making inactivated poliovirus vaccines. Yes, yes, what some conspiracy theorists claim happened in the «mysterious» virology lab in Wuhan has happened before in Europe itself, without the western media or any crazy president pointing an accusing finger at us.
.
But inactivated SARS-CoV-2 vaccines, unlike mRNA and other technologies widely supported by Operation Warp Speed, have a strong track record. «There are many different ways to make vaccines, and it's great that innovation is happening alongside proven approaches,» says Nicole Lurie, strategic advisor to the Coalition for Epidemic Preparedness Innovation (CEPI), who previously served as US assistant secretary for preparedness and response. «Inactivated vaccines are one of several proven approaches.» Meng Weining, senior director of Sinovac, says they compared the inactivated approach - which they already use to make six vaccines - with two other strategies in animal models. «The inactivated whole virus vaccine gave a much, much better result,» says Meng.
.
Although it is theoretically easier to produce mRNA in large quantities than to grow the virus on a similar scale, vaccine experts say the production of inactivated virus vaccines is unlikely to be an obstacle. CNBG, for example, has «enormous resources»: 10,000 employees and scientists, huge manufacturing capacity,» says Nicholas Jackson, who heads CEPI's China office and formerly worked in vaccine R&D at Pfizer. «They are a very competent beast.» And, crucially for Chinese vaccine diplomacy, many other countries have manufacturers that have been producing inactivated virus vaccines for decades.
.
If China's COVID-19 vaccines work, manufacturers say they could produce 1.5 billion doses in total next year. And countries that cannot access Warp Speed-funded vaccines, especially those that have hosted Chinese efficacy trials, could have a more assured vaccine supply.
.
Sheikh Mohammed Bin Rashid Al Maktoum, prime minister of the United Arab Emirates (UAE), on 3 November tweeted a photo of himself in Dubai with the right sleeve of his kandura rolled up high, being injected with a CNBG COVID-19 vaccine. «We wish everyone safety and great health, and we are proud of our teams who have worked tirelessly to make the vaccine available in the UAE,» Al Maktoum wrote. Two of the country's top ministers had received the vaccine three weeks earlier.

Pictured above is Mohammed bin Rashid Al Maktoum (left), Prime Minister of the United Arab Emirates, shortly after receiving a COVID-19 vaccine from CanSino Biologics on 3 November under his country's emergency use authorisation.
.
The UAE has become the cornerstone of the NBSC efficacy trials and is following China's controversial lead in allowing people to receive the vaccine outside of clinical trials.
.
At a 23 June videoconference linking Abu Dhabi, Beijing and Wuhan, health officials, ambassadors and CNBG executives sat at long tables in rooms decorated with the flags of each country and celebrated their decision to conduct a trial together to assess efficacy. The trial has since expanded to Bahrain, Egypt and Jordan and is expected to recruit 45,000 people. CNBG says it came to the UAE to test its two whole-virus vaccines - similar inactivated preparations made by two independent, and even competing, laboratories, one in Wuhan and the other in Beijing - because the high rate of SARS-CoV-2 infection there should hasten a sign of efficacy. But diplomacy and trade also drove the decision. The UAE's huge foreign workforce means that trial participants come from 125 different countries. «If you can prove that these vaccines work in the UAE,» says Huang, «that means everyone would think the vaccine would work in their countries too.».
.
China may be hoping for a public relations (PR) benefit as well: The United Arab Emirates and many of the other partner countries have large Muslim populations, which Huang says could help mitigate human rights complaints about China's treatment of Uighur Muslims in Xinjiang province. «They certainly don't want to have more enemies abroad,» he says.
.
Huang adds that through its series of overseas trials, China hopes to create goodwill for its Silk Belt and Road Initiative (BRI), massive infrastructure investment in more than 100 countries to increase trade. Critics have accused the BRI of being «debt trap» diplomacy that is a form of neo-colonialism. «China wants to work with these countries and prioritise them to have the vaccine because I think this will facilitate the implementation of the BRI,» he says.
.
China's vaccine diplomacy has not always worked well. On 9 November, after Brazil suspended a Sinovac vaccine trial following the death of a participant, President Jair Bolsonaro took to Facebook. «Death, disability, anomalia,» he wrote, quoting a Brazilian health agency that had listed the possible reasons for the suspension: death, disability, genetic anomalies. Bolsonaro's message was clear: This Chinese vaccine, called CoronaVac, was dangerous.
.
«A lot of people were very surprised by that post,» says Esper Kallas, who runs the vaccine trial centre at the University of São Paulo that the participant had joined. «He was celebrating the failure of a vaccine.» For Bolsonaro, it was a public relations victory over his political archenemy, the governor of São Paulo, who supported the CoronaVac trial. The president was also reveling in a setback for China, which Bolsonaro, like his ally, US President Donald Trump, has relentlessly criticised.
.
It turned out that the participant died of a drug overdose. His death had nothing to do with the CoronaVac, and the trial was quickly resumed.
.
China chose to navigate Brazil's daunting politics because with an out-of-control pandemic - it is third in the world in total infections, with more than 100,000 new cases every week - the country is a magnet for vaccine trials and is desperate for vaccines. The state of São Paulo in September committed $90 million to Sinovac for 46 million doses (this is 10 times cheaper than what the US government is paying for mRNA vaccines from Pfizer/BioNTech and Moderna). And Brazil could increase supply by manufacturing the vaccine on site under licence. Sinovac says it could transfer its technology to Instituto Butantan, a major vaccine manufacturer in Sao Paulo, a collaboration Meng describes as «win-win».
.
China has had warmer receptions in other countries. In September, Turkey launched a 13,000-person efficacy trial of Sinovac's vaccine. Serhat Ünal, who heads Hacettepe University's Vaccine Institute - which is similar to Butantan's in Brazil - and sits on the health ministry's scientific board, says Turkey has «a good infrastructure for phase III studies» and, unlike the US and much of Europe, hosted a Chinese vaccine manufacturer.
.
The three Chinese manufacturers also have large efficacy trials planned or underway in Indonesia, Pakistan, Saudi Arabia, Mexico and Chile (see map above). It's a good strategy, says Ünal. «When you do phase III in different countries, it's more transparent, it's more reliable,» he says.
.
As much as vaccine diplomacy influences the deals Chinese vaccine manufacturers make for efficacy trials, they are also driven by capitalism, says Yip, who for four years headed the China office of the US Centers for Disease Control and Prevention (CDC). «Everyone is clamouring for a COVID vaccine,» he says. «Everyone wants to tell their people that we've got some vaccine for you.» Y Chinese companies will make profits when supplying it.
.
It is a safe bet that one or more of China's overseas trials will announce efficacy data any day now. Results so far from other vaccines have fuelled a growing sense that many vaccines will crush, what is, from a vaccine's point of view, a somewhat weak virus. But China is not waiting for phase III results before using vaccines widely at home. Its regulators appear to be satisfied with the animal studies combined with minimal safety and immune response data from phase I and II trials. In June, CanSino received an emergency use authorisation to vaccinate the military, and since then both Sinovac and CNBG have been given the green light to vaccinate large populations outside of clinical trials. Unconfessable but presumably very significant numbers.

A refrigerated container with a batch of 120,000 doses of Sinovac's Chinese COVID-19 vaccine arrived at São Paulo international airport on 19 November. The vaccine will be used if ongoing efficacy trials show it to be safe and effective.
.
With the pandemic defeated at home, China is vaccinating its people as insurance, often against a dangerously infected world.. CanSino's Yu says «thousands» of troops on peacekeeping missions have received his company's vaccine before travelling to places with a high burden of COVID-19. CNBG says «hundreds of thousands» of people in China have received their vaccines. «By doing this, we are able to build an immunological barrier among specific groups of people such as health care workers, pandemic prevention personnel and border inspection personnel,» the company explained in its written responses to Science. Vaccination is «completely voluntary with informed consent,» CNBG stresses. Moreover, «We did not receive a single case report of a severe adverse reaction, and no infections were reported for vaccinees working in high-risk areas».
.
Sinovac's Meng says that «more than 90%» of the company's employees have received their vaccination because they are considered a high-risk group; he received it because he travels abroad. (According to China's Ministry of Culture and Tourism, 155 million Chinese travelled abroad in 2019, including thousands of students university students who are continuing their education in the best American universities). In October, the company began selling its vaccine -$60 for two doses - in Yiwu, a city in Zhejiang province. And Yip says the government was even considering vaccinating all of Beijing after an outbreak of COVID-19 there in June. Yip says officials «had already written the guidelines»; if more than 500 cases had come to light, «they would inject everyone in Beijing with the vaccine». In the end, contact tracing, testing and isolation of infected people limited the outbreak to 335 cases.
.
In the end, contact tracing, testing and isolation of infected persons limited the outbreak to 335 cases.
.
Morrison says that the Chinese government has «clearly decided at the highest level» that it is worth betting on creating «facts on the ground»and gain a global commercialisation advantage by having the first COVID-19 vaccines in wide use. «It's a risk but it's also potentially a big win,» he says.
.
But what happens if damage is done? «You shouldn't apply peacetime rules during war. Our lives are turned upside down,» says Yip, who lives part-time in Beijing.
.
If its range of vaccines is successful, China's image will gain a boost both at home and abroad. «They have reputational problems, internally and externally,» says Morrison.
.
In May, China's President Xi Jinping told the World Health Assembly, which governs the World Health Organisation (WHO), that the country would make its COVID-19 vaccines «a global public good», a somewhat vague statement that left many China watchers scratching their heads. However, China fulfilled this commitment in October by joining the COVID-19 Global Vaccine Access Service (COVAX)., The WHO and UNECE are leading an effort in part to ensure that any product that is proven safe and effective reaches rich and poor countries alike quickly.
.
This is primarily a diplomatic move. COVAX has not received support from the US or Russia, and China sees that it «could have a controlling influence over an important international mechanism». Moreover, says Alexandra Phelan, a lawyer at Georgetown University's Center for Global Health Science and Security who specialises in China, «It is a good act of a global citizen to support this effort».
.
If a vaccine made in China proves safe and effective, it could help people forget the pandemic that started there and how poorly the government responded at first, says Morrison. And at home, it could absolutely clean up the image of China's vaccine manufacturers. Chinese citizens have recovered from a series of scandals over the past decade that include the use of ineffective vaccines against diphtheria, pertussis and tetanus; inadequate registrations of a rabies vaccine; and sales of an expired polio vaccine.
.
A successful Chinese-made COVID-19 vaccine that has been vetted by external regulators would reassure the domestic market, says Phelan. «There is a lot of domestic ground to make up.».
.
In Brazil, Kallas says a similar dilemma could arise if Butantan, as expected, starts manufacturing Sinovac's CoronaVac. «There is a saying here that the neighbour's chicken is always the tastiest,» says Kallas. «We have a perception that everything we make is not as good as the imported stuff.»
.
But for now, Brazil is embracing the Chinese vaccine. With cases on the rise, the arrival of just 120,000 doses of CoronaVac on 19 November became big news. The bias against China is little more than a far-right political «contamination», says Kallas, and most Brazilians see CoronaVac as «a viable option».
.
In short, China is managing its vaccine supply trials around the world intelligently and strategically, with no epidemiological fires to put out within its borders. As always, the Chinese are the smartest in the class. And to their economic clout they are now adding biological and diplomatic clout. Soon no country in the world will be able to afford to be China's enemy. And its businesses of all kinds are there, freely quoted and available to any investor.